Researchers Develop New Method for Biomanufacturing Vascularized Tissue
Date:03-02-2021 | 【Print】 【close】
In native tissues and organs, vascularized structures infiltrated within the natural nanofibrous extracellular matrix (ECM) play vital roles in maintaining the functions and metabolisms of tissues and organs. However, restricted by existing strategies, the vascularization of synthetic ECM-mimicking nanofibrous tissue engineering scaffold remains greatly challenging, because three-dimensional (3D) infiltration and incorporation of vascular endothelial cells within nanofibrous scaffolds is extremely difficult.
To address this challenge, Dr. ZHAO Qilong’s group from the Shenzhen Institute of Advanced Technology (SIAT) of the Chinese Academy of Sciences collaboration with Prof. WANG Min’s group at the University of Hong Kong reported a new method for biomanufacturing vascularized tissues.
The study was published in Acta Biomaterialia on Jan 27.
The team developed a new biomanufacturing method of concurrent emulsion electrospinning and coaxial cell electrospraying for simultaneously depositing cell-embedded hydrogel microspheres along with vascular endothelial growth factor (VEGF)-containing nanofibers, subsequently realizing 3D endothelial cell incorporation in electrospun nanofibrous scaffolds independent of cell penetration after selectively dissolving the hydrogel microspheres for releasing the endothelial cells.
To identify the viability of endothelial cells in the scaffolds formed by the concurrent biomanufacturing method, the level of reactive oxygen species (ROS) for the cells in the hydrogel microspheres after the cell electrospraying and in the scaffolds after the cell release treatment have been monitored, indicating that there are no significantly changes in compared to the cells normally cultured on the scaffolds.
Furthermore, the researchers found that under the inducements of combined structural and biochemical cues via the 3D cell-incorporated architectures, endothelial cells could freely stretch, display enhanced intercellular connections, and maintain the phenotype in the bioactive nanofibrous scaffolds, implying improved vascularization potential.
This study offers a promising platform technology for creating bioactive nanofibrous scaffolds with 3D cell incorporation and for overcoming inherent problems of nanofibrous tissue engineering scaffolds, which should open new avenues for biomanufacturing tissue-mimicking constructs with vascularized structures.
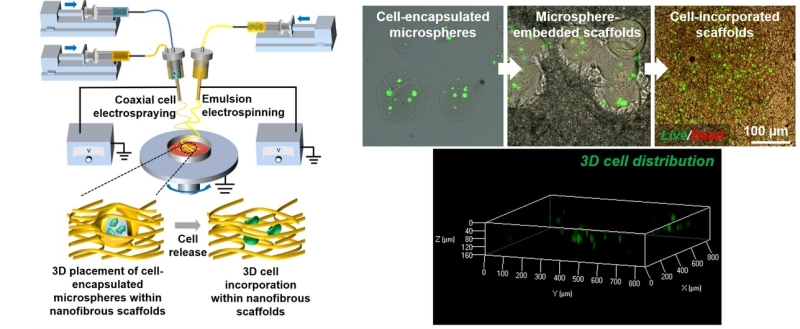
A schematic diagram showing the concurrent emulsion electrospinning and coaxial cell electrospraying process for fabricating firstly nanofibrous scaffolds embedded with cell-encapsulated microspheres and subsequently nanofibrous scaffolds with 3D cell incorporation after the release of encapsulated cells from microspheres. (Image by SIAT)
Media Contact:
ZHANG Xiaomin
Email: xm.zhang@siat.ac.cn