Scientists Erase Drug-Associated Memories to Prevent Drug-Associated Memory
Date:17-07-2020 | 【Print】 【close】
Research surrounding addiction often points to reward as the primary motivation for drug use and relapse. But while chasing a "high" can initiate drug use, it’s often the acute symptoms of withdrawal — which can include nausea, vomiting, pain and cramping — that drives a return to drugs for relief.
The most difficult part of treating addiction is to prevent relapse, especially for opioids, opioid withdrawal symptoms are severe and relapse among users is common.
Both the reward of the drug "high" and the alleviation of painful withdrawal symptoms can serve as powerful memory cues that trigger a relapse.
Researchers from the Shenzhen Institutes of Advanced Technology (SIAT) of the Chinese Academy of Sciences and Stanford University interrupted the brain pathway responsible for morphine-associated memories in mice, so to speak, "erasure" the drug-associated memory from the brain.
The study was published in Neuron on July 16, 2020.
Firstly, researchers introduced the mice to a two-sided chamber for training, differentiated by tactile and visual cues. On one side they were given saline, on the other side a small dose of morphine. After 5 days, the animals had unsurprisingly developed a compulsive preference for the chamber with morphine. By then, the mice had become addicted.
And then, by using light from an optical fiber, the scientists could turn the paraventricular thalamus (PVT) pathway off, to alleviate opiate withdrawal symptoms, which was found by the same team in 2016.
When the withdrawal pathway from the PVT was turned off or silenced, the mouse’s preference for the drug-associated chamber went away. If tested a day later — when the withdrawal pathway could theoretically function again and environmental cues could reactivate the memory — the scientists were surprised to find that the mice still showed no preference for the drug-associated chamber.
"Our data suggest that after silencing this PVT pathway, environmental cues will not work to reactivate this memory," said CHEN Xiaoke, an associate professor of biology at Stanford University.
Even when morphine was reintroduced to the chamber, the mice still did not preferentially go there, and this held true even two weeks later. It’s as if the animals had completely forgotten the effects — both good and bad — of the drug.
"Our success in preventing relapse in rodents may one day translate to an enduring treatment of opioid addiction in people," said ZHU Yingjie, the co-correspondence author of this study from SIAT.
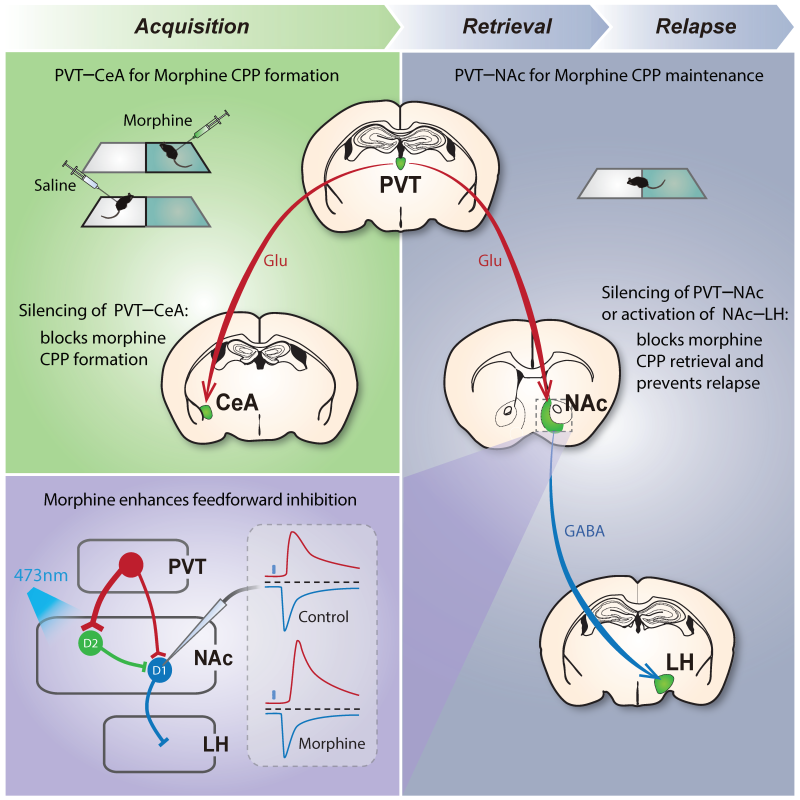
Figure. The PVT/CeA pathway associates opiate reward to the environment, whereas transient manipulation of the PVT/NAc pathway or its downstream NAc/LH pathway during retrieval erases opiate-associated memory and causes enduring protection against relapse to opioid use. (Image by SIAT)
Media Contact:
ZHANG Xiaomin
Email: xm.zhang@siat.ac.cn